See also
Watch the webinars on the SPHN Framework and its Toolstack and the SPHN Data Ecosystem for FAIR Data!
Introduction
Personalized health research relies on access to diverse health data. However, this data is often scattered across different sources (i.e., hospitals, laboratories, or mobile devices), and stored in various formats, making it challenging to collect, connect and analyze effectively.
To address this challenge, the Swiss Personalised Health Network (SPHN) initiative was launched in 2017 with the support from the State Secretariat for Education, Research, and Innovation (SERI) in Switzerland.
Today, SPHN operates as a national data infrastructure under the responsibility of the Swiss Academy of Medical Sciences (SAMS) in collaboration with the SIB Swiss Institute of Bioinformatics (SIB).
SPHN’s mission is to make health data interoperable and shareable for research in Switzerland, bridging the gap between data providers (such as hospitals and healthcare institutions) and researchers.
SPHN Semantic Interoperability Strategy
SPHN provides a framework for exchanging health-related data to ensure interoperability, meaning data from different sources can be seamlessly integrated, compared, and used together for research.
Current Challenges
While some coding systems exist for patient billing and accounting, Switzerland currently lacks widely adopted standards for medical data in hospitals.
SPHN’s solution
To enable the use of health-related data from clinical routine and other sources for research, SPHN has developed a semantic interoperability strategy [Gaudet-Blavignac et al. 2021], based on the FAIR principles and reusing existing standards.
This strategy is built on three pillars:
SPHN Concepts: Semantics defining meaning and relationships of the data
SPHN RDF Schema: Standardized data transport and storage
Use cases: demonstrates practical applications of the framework
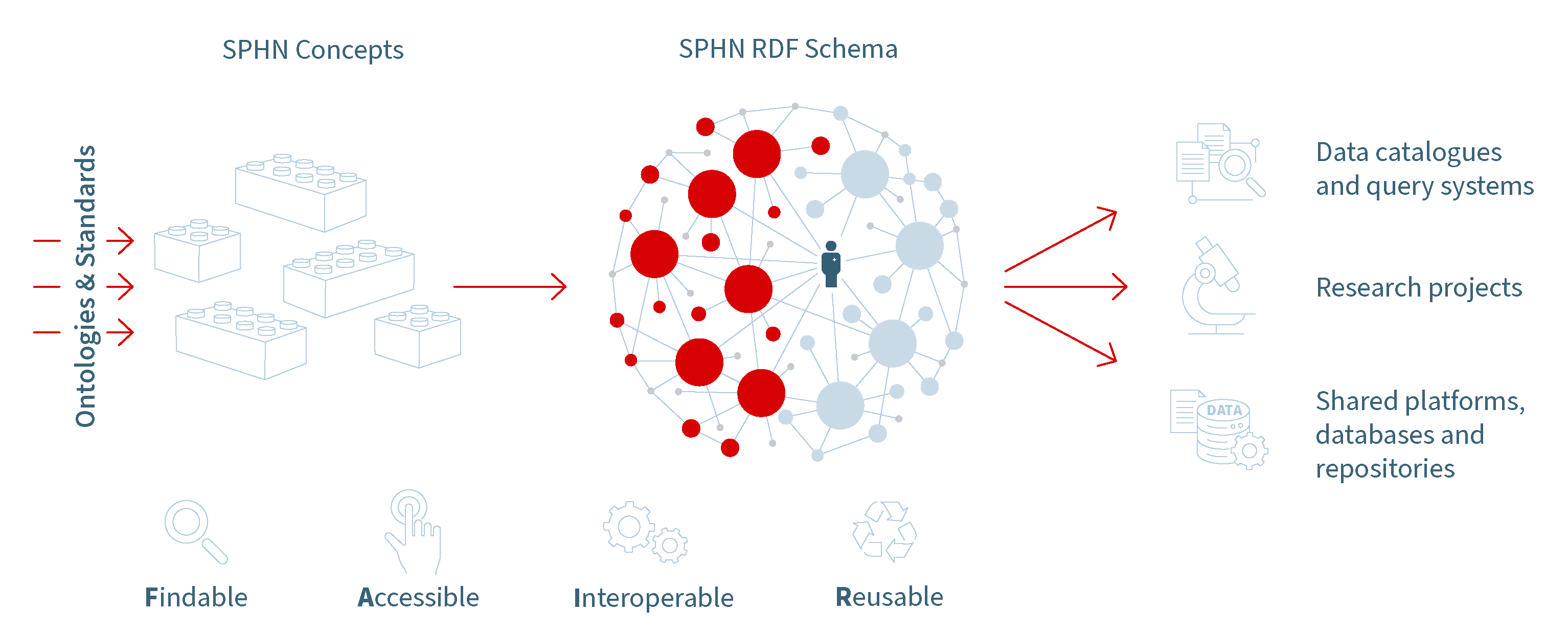
Figure 1. The Three Pillars of SPHN’s Semantic Interoperability Strategy - A framework for seamless data integration in research.
Stakeholder Roles
SPHN fosters a structured and collaborative environment where data producers and consumers work together to advance personalized health research in Switzerland.
Role |
Who? |
What do they do? |
|---|---|---|
Data Producer |
Clinical Data Manager |
Collect, organize, and maintain clinical data in hospitals or clinics. Their job is to make sure this data is ready and available for researchers to use |
Data User |
Project Data Manager |
Technical expert who prepares and adapts data for specific research projects |
Data User |
Researcher |
Scientist who uses the data to study and find answers to biomedical questions. |
Data User |
Project Leader |
In charge of a research project, making sure the project stays on track. |
These groups work together as part of the SPHN Ecosystem.
SPHN implementation teams coordinate the building and maintenance of infrastructures to support these stakeholders.
SPHN Core Ecosystem
The SPHN initiative has built an ecosystem design to simplify and secure the sharing and reuse of health data for researchers and healthcare providers [Österle et al. 2021] . This ecosystem includes key components that standardizes how data is represented and exchanged:
As part of Pillar 1, the SPHN Dataset, defines the meaning of the data (i.e. semantics) for consistency across research and clinical settings. For example, it specifies what information is needed to describe an allergy, ensuring everyone uses the same terms and definitions. The SPHN Dataset provides a standardized way to describe health-related concepts used in Swiss health research.
The semantics defined in the SPHN Dataset are then structured using RDF (Resource Description Framework) (Pillar 2). This ensures data providers can share clinical data in a consistent and standardized format aligned with the FAIR principles. These principles, promote making data Findable, Accessible, Interoperable and, Reusable across diverse sources. The outcome of this standardization is the SPHN RDF Schema, which provides a clear, organized framework for data exchange.
To ensure compatibility with national and international terminologies, SPHN developed a Terminology Service. This automatically converts existing health-related standards into the RDF format, making them accessible to stakeholders within SPHN. By reusing these standards, SPHN ensures data consistency within Switzerland but also enhances compatibility with international systems, making data more reliable, shareable and comparable across sources.
SPHN Data Journey for FAIR Data
Project initiation
At the start of a project, the first step is to define the scope and assess whether the study is feasible. To help with this, the metadata of the SPHN Federated Clinical Routine Dataset provides valuable insights into the available datasets from participating hospitals across the SPHN network. This allows to evaluate the data landscape and determine if the necessary data exists to support the research goals.
Once feasibility is confirmed, all involved parties must finalize and sign a legal agreement to formally initiate the project (more information on legal aspect can be found here).
Extending semantics and schemas
Scientific projects have the possibility to extend the semantics defined in the SPHN Dataset and SPHN RDF Schema to include project-specific information.
This extension can be achieved in two ways:
Projects can represent their semantics in the SPHN Dataset Template and uses the SPHN Schema Forge webservice to automatically generate the project-specific RDF Schema.
Projects can directly build their schema in RDF format using their preferred editor.
Once the Project Data Manager finalizes a project-specific RDF Schema, they send it to the data providers.
Data preparation
Data providers then integrate this schema into their pipelines to transform data from clinical data warehouses and generate RDF data files compliant with the schema. Since 2023, data providers can use the SPHN Connector to create SPHN-compliant and valid RDF data efficiently.
Any new data generated by a clinical data manager undergoes automated quality checkes integration in the SPHN Connector. This tool integrates the Quality Check tool, which uses the SHACLer and SPARQL queries to ensure data integrity against the provided schema.
Once data is validated, researchers can explore and analyze it according to their needs in BioMedIT, a secure IT network and trusted research environment for responsibly processing health data. For more details on data transfers and usage within BioMedIT, visit biomedit.ch.
Data Reuse
The SPHN Metadata Catalog references datasets available in the SPHN network. This catalog enables researchers to easily find existing datasets for reusability (Pillar 3).
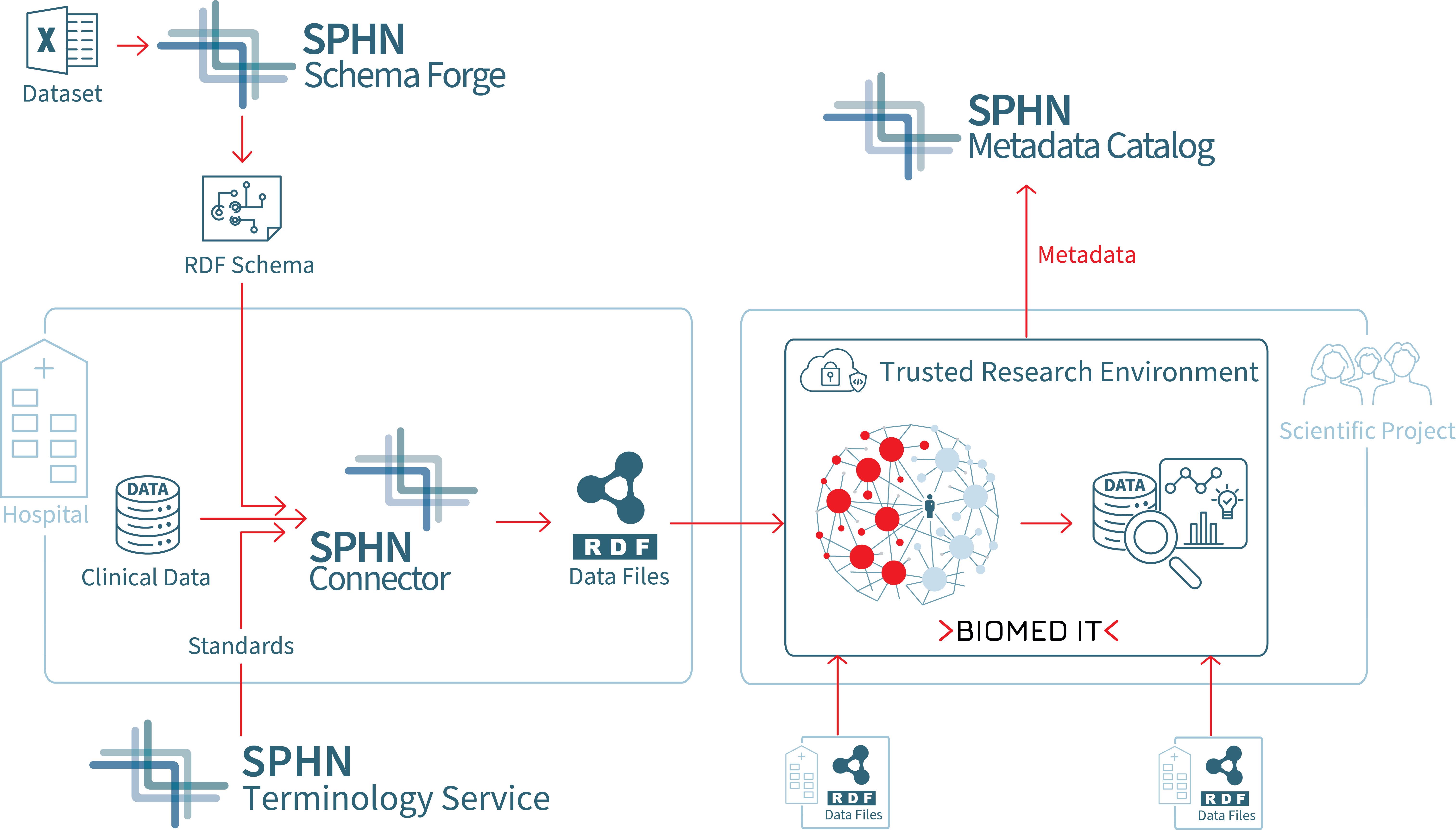
Figure 2. Simplified overview of the SPHN Ecosystem. This diagram provides a streamlined workflow of the SPHN Semantic Interoperability Framework highlighting key steps for hospitals and scientific project members. It showcases how data is standardized, shared, utilized, and made findable within the SPHN network for research purposes.
Reference
Österle, S.; Touré, V.; Crameri, K. (2021), The SPHN Ecosystem Towards FAIR Data. Preprints, 2021090505 (doi: 10.20944/preprints202109.0505.v1)
Gaudet-Blavignac C, Raisaro JL, Touré V, Österle S, Crameri K, Lovis C (2021), A National, Semantic-Driven, Three-Pillar Strategy to Enable Health Data Secondary Usage Interoperability for Research Within the Swiss Personalized Health Network, JMIR Med Inform 9 (6), e27591 (doi: 10.2196/27591)
Touré, V., Krauss, P., Gnodtke, K., Buchhorn, J., Unni, D., Horki, P., Raisaro, J.L., Kalt, K., Teixeira, D., Crameri, K. and Österle, S. (2023), FAIRification of health-related data using semantic web technologies in the Swiss Personalized Health Network. Scientific Data, 10(1), p.127 (doi: 10.1038/s41597-023-02028-y)
Armida, J., Touré, V., Krauss, P., Unni, D., Witte, H., Chiarugi, D., Marto, A.B., et al. (2025), Semantic Interoperability at National Scale: The SPHN Federated Clinical Routine Dataset., Research Square (preprint) (doi: 10.21203/rs.3.rs-8250886/v2)
See also
If you’re new to Semantic Web technologies, we recommend reading Background Information section of this document before continuing.